The concept of chemical equilibrium and reacting systems
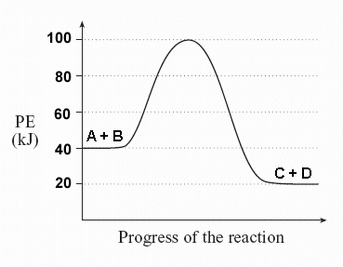
Q. How most chemical reactions are reversible?
A. The tendency of a chemical reaction is to go to the lowest energy state. However, since there is still energy in system, which can give sufficient amount of energy for both reactants and product, the reactants can go over the activation energy to form the products and vice versa. That's why most chemical reactions are reversible.
A. The tendency of a chemical reaction is to go to the lowest energy state. However, since there is still energy in system, which can give sufficient amount of energy for both reactants and product, the reactants can go over the activation energy to form the products and vice versa. That's why most chemical reactions are reversible.
<Fig.1 - a potential energy diagram>
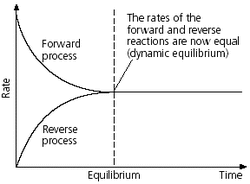
Q. Describe the dynamic nature of chemical equilibrium.
A. In a state of equilibrium, the rates of forward reaction and backward reaction become the same (reach the same level). Thus, at chemical equilibrium, it seems there is no change (macroscopic or invisible) in the system. However, the reactions still happen in the system, which is the reason why it is called as 'the dynamic equilibrium'.
<fig 2 - rates graph at equilibrium>
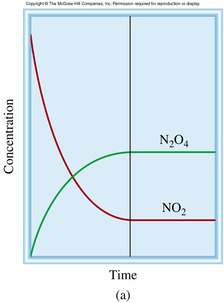
Q. explain and provide an example that
shows that a system not at equilibrium will tend to move toward a position of
equilibrium
A. Let's say N2O4 is put in an empty system, and N2O4 can reach at equilibrium in this following equation : N2O4 ⇋ 2NO2. Since N2O4 is the only things in the system, the rate of forward reaction is fast. As the reaction proceeds, NO2 is formed and reverse reaction begins. In the meantime, the rate of forward reaction decreases because [N2O4] decrease, besides the rate of reverse reaction increases because [NO2] increases. Ultimately, both reaction rates (forward and reverse) reach at the same speed, which shows the system has reached at equilibrium.
A. Let's say N2O4 is put in an empty system, and N2O4 can reach at equilibrium in this following equation : N2O4 ⇋ 2NO2. Since N2O4 is the only things in the system, the rate of forward reaction is fast. As the reaction proceeds, NO2 is formed and reverse reaction begins. In the meantime, the rate of forward reaction decreases because [N2O4] decrease, besides the rate of reverse reaction increases because [NO2] increases. Ultimately, both reaction rates (forward and reverse) reach at the same speed, which shows the system has reached at equilibrium.
<fig 3 - the graph of [N2O4] and [NO2]>
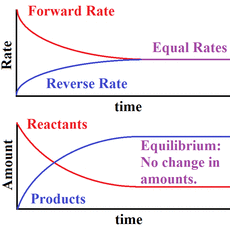
Q. describe chemical equilibrium as a closed system at constant temperature that has the following conditions :
- macroscopic properties are constant, -the forward and reverse reaction rates are equal, - can be achieved from either direction, -the concentrations of reactants and products are constant
<fig.4 - the graph of the rates and the concentrations at equilibrium>
A. If the system remains at constant temperature, and in a closed system, macroscopic is meant to be constant at chemical equilibrium, because the rates of forward reaction and reverse reaction are the same; it seems there is no change in the system at all: no change in the concentrations of reactant and product, total pressure, colour, temperature, density, etc. The forward and reverse reaction rates are also equal, since there is no change in the system at equilibrium; if there is any change in the system at equilibrium, it means that the rates of forward and reverse reaction are not the same. Moreover, equilibrium can be achieved from either direction because as we have seen it above, a system that is not at equilibrium will tend to move toward a position of equilibrium, no matter from which side you start the reaction. Furthermore, the concentrations of reactants and products are constant, for the same reasons as before: the rates of forward reaction and reverse reaction are the same, which means there is no change at all.
- macroscopic properties are constant, -the forward and reverse reaction rates are equal, - can be achieved from either direction, -the concentrations of reactants and products are constant
<fig.4 - the graph of the rates and the concentrations at equilibrium>
A. If the system remains at constant temperature, and in a closed system, macroscopic is meant to be constant at chemical equilibrium, because the rates of forward reaction and reverse reaction are the same; it seems there is no change in the system at all: no change in the concentrations of reactant and product, total pressure, colour, temperature, density, etc. The forward and reverse reaction rates are also equal, since there is no change in the system at equilibrium; if there is any change in the system at equilibrium, it means that the rates of forward and reverse reaction are not the same. Moreover, equilibrium can be achieved from either direction because as we have seen it above, a system that is not at equilibrium will tend to move toward a position of equilibrium, no matter from which side you start the reaction. Furthermore, the concentrations of reactants and products are constant, for the same reasons as before: the rates of forward reaction and reverse reaction are the same, which means there is no change at all.
