Cu(s) + 4HNO3(aq) --> Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) + heat
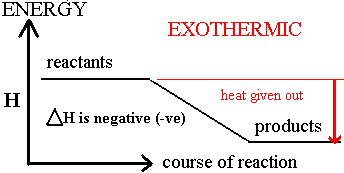
Above you can see how the reactants have the least amount of randomness as there is one solid and one aqueous solution while on the product side there is no solid but an aqueous solution, a pure liquid, and a gas. There for the product side has the tendency to strive for maximum entropy (randomness). On the other hand the side with the heat term ( product side ) has the tendency to lean towards minimum enthalpy.
Above you can see how the reactants have the least amount of randomness as there is one solid and one aqueous solution while on the product side there is no solid but an aqueous solution, a pure liquid, and a gas. There for the product side has the tendency to strive for maximum entropy (randomness). On the other hand the side with the heat term ( product side ) has the tendency to lean towards minimum enthalpy.
If both the the entropy and enthalpy factors favor the products the reaction will go 100%, meaning the reactants will be all used up and there will only be products left over.
Cu(s) + 4HNO3(aq) --> Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) + heat
If both the entropy and enthalpy factors favor the products the reaction will not occur at all.
Heat + Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) --> Cu(s) + 4HNO3(aq)
If the entropy and enthalpy factors oppose each other the reaction will form an equilibrium.
Cu(s) + 4HNO3(aq) --> Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) + heat
Cu(s) + 4HNO3(aq) --> Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) + heat
If both the entropy and enthalpy factors favor the products the reaction will not occur at all.
Heat + Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) --> Cu(s) + 4HNO3(aq)
If the entropy and enthalpy factors oppose each other the reaction will form an equilibrium.
Cu(s) + 4HNO3(aq) --> Cu(NO3)2(aq) + 2H2o(l) + 2NO2(g) + heat