Le Châtelier’s principle
Q. explain the term shift as it applies to an equilibrium system
A. In an equilibrium system, 'shift' (counteraction) occurs when a closed system at equilibrium gets a stress (disturbance) that changes a state of equilibrium, in order to counteract the stress (disturbance). It is also what Le Chatelier's principle means to say.
A. In an equilibrium system, 'shift' (counteraction) occurs when a closed system at equilibrium gets a stress (disturbance) that changes a state of equilibrium, in order to counteract the stress (disturbance). It is also what Le Chatelier's principle means to say.
<fig.1 the graph of the changes when a stress is given in an equilibrium>
Q. describe how a change in the following factors (stressors) causes a shifts in an equilibrium system:
-temperature change (exothermic and endothermic), -concentration change, -volume change of gaseous systems
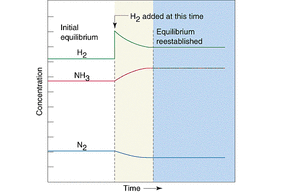
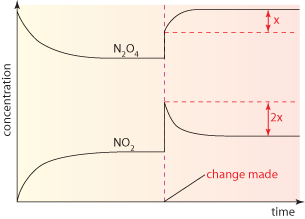
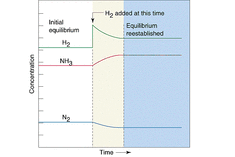
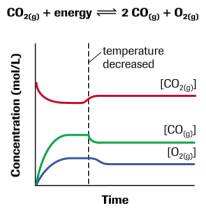
A. According to Le Chatelier's Principle, processes will occur that tend to counteract that change. Thus, if there is a temperature change, processes will occur that tend to recover the temperature change. For instance, in exothermic reaction, if temperature decreases, processes will occur that tend to increase the temperature. Therefore, the forward reaction rate will increase, consequently, the equilibrium will shift toward the right side. if it is endothermic reaction, if temperature increases, processes will occur that tend to decrease the temperature. Therefore, the reverse reaction rate will increase, consequently, the equilibrium will shift toward the left side. Suppose if there is a concentration change, then according to Le Chatelier's Principle, processes will occur that tend to recover the concentration change. Given an equilibrium system, and in the system there is some molecules that comply a following equation: A + B ⇋ C. If a concentration of A is increase, according to the principle, process will occur that tend to lower the concentration of A. Therefore, the forward reaction rate will increase in order to decrease the concentration of A. Thus, the equilibrium will shift toward the right side. For a volume change of gaseous systems, if there is a change in volume, which is equal to a change in pressure (yet they are opposite proportional), processes will occur that tend to recover the volume change. Let's say there is an equilibrium system that has two kinds of gaseous molecules that comply the following equation : C(g) ⇋ 2D(g). If the pressure of system increases, which means the volume of system decreases, processes will occur that tend to decrease the pressure of the system. Thus, the reverse reaction rate will increase, since D has bigger coefficient than C. As a result, the equilibrium will shift to left side.
-temperature change (exothermic and endothermic), -concentration change, -volume change of gaseous systems
A. According to Le Chatelier's Principle, processes will occur that tend to counteract that change. Thus, if there is a temperature change, processes will occur that tend to recover the temperature change. For instance, in exothermic reaction, if temperature decreases, processes will occur that tend to increase the temperature. Therefore, the forward reaction rate will increase, consequently, the equilibrium will shift toward the right side. if it is endothermic reaction, if temperature increases, processes will occur that tend to decrease the temperature. Therefore, the reverse reaction rate will increase, consequently, the equilibrium will shift toward the left side. Suppose if there is a concentration change, then according to Le Chatelier's Principle, processes will occur that tend to recover the concentration change. Given an equilibrium system, and in the system there is some molecules that comply a following equation: A + B ⇋ C. If a concentration of A is increase, according to the principle, process will occur that tend to lower the concentration of A. Therefore, the forward reaction rate will increase in order to decrease the concentration of A. Thus, the equilibrium will shift toward the right side. For a volume change of gaseous systems, if there is a change in volume, which is equal to a change in pressure (yet they are opposite proportional), processes will occur that tend to recover the volume change. Let's say there is an equilibrium system that has two kinds of gaseous molecules that comply the following equation : C(g) ⇋ 2D(g). If the pressure of system increases, which means the volume of system decreases, processes will occur that tend to decrease the pressure of the system. Thus, the reverse reaction rate will increase, since D has bigger coefficient than C. As a result, the equilibrium will shift to left side.
<fig 2, 3, 4 (in order). the graph of changes in concentrations when temperature, concentration, pressure is changed>
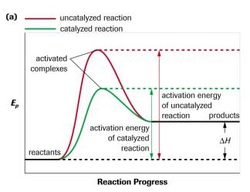
Q. describe the effect of a catalyst on dynamic equilibrium
A A. Since a catalyst lower the activation energy that affect either forward and reverse reaction, the rate of both reactions increase by the same amount. Therefore, the equilibrium will not shift.
A A. Since a catalyst lower the activation energy that affect either forward and reverse reaction, the rate of both reactions increase by the same amount. Therefore, the equilibrium will not shift.
<fig 4. the graph of the activation energy of uncatalyzed reaction and catalyzed reaction>
Q. use examples to explain how the reaction rates and concentrations of an equilibrium system are changing when a stress is applied
A. if you refer to Q. describe how a change in the following factors (stressors) causes a shifts in an equilibrium system (above), and fig 2,3,4, it would help for this question.
A. if you refer to Q. describe how a change in the following factors (stressors) causes a shifts in an equilibrium system (above), and fig 2,3,4, it would help for this question.